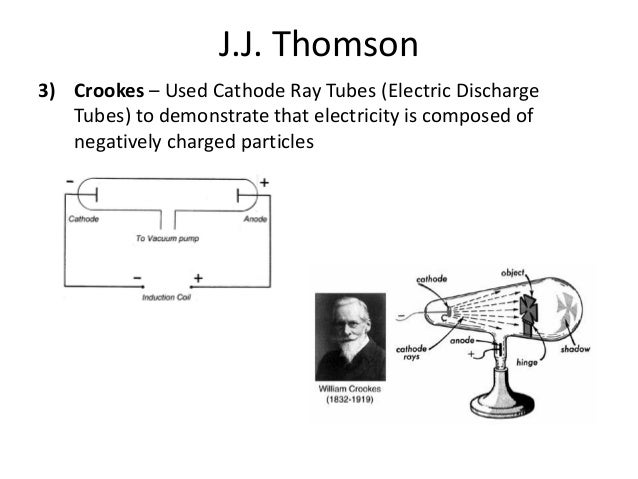
To identify the constituents of the ray produced by applying a high voltage to the tube, the dipole was set up as an add-on in the experiment.ĥ. The electricity starts flowing as the circuit was complete.Ĥ. High voltage is passed to the two metal pieces to ionize the air and make it a conductor of electricity.ģ. Apparatus is set up by providing a high voltage source and evacuating the air to maintain the low pressure inside the tube.Ģ. Instead, what flowed off the cathode toward the anode were called ‘cathode rays.’ Hence the name cathode ray tube. So, no one was calling it an electron beam. Before his experiment, we didn’t know electrons existed. Thomson to see where the electron beam was hitting. On the opposite side of the tube is a coating that glows when struck by the electrons. A small hole in the anode allows some electrons to pass through it, creating a beam of electrons. Electrons, which have a negative charge, flow off the cathode and are attracted towards the anode. The cathode is a negatively charged conductor, and the anode is a positively charged conductor. On one side of the inside of the tube, there’s a cathode and an anode. The CRT consists of several elements, starting with a vacuum-sealed tube to keep air out of it. As seen in the diagram below, we’ll look at a basic CRT like what J.J. Thomson used the cathode ray tube to discover the electron, we need to know how a cathode ray tube works. Working of Cathode Ray Tubesīefore we see how J.J. The pressure of the gas inside the tube was lowered by evacuating the air.

The two metal pieces were connected with an external voltage. The experiment apparatus incorporated a tube made of glass containing two pieces of metals at the opposite ends which acted as an electrode. He was able to calculate the charge-to-mass ratio of the electron.Īfter these three experiments, he deduced that inside the atom, there consists a subatomic particle, named initially ‘corpuscle,’ then changed to ‘electron,’ which is 1800 times lighter than the mass of hydrogen atom (Lightest atom).

This experiment concluded that the electrostatic deflection is the same as the electromagnetic deflection for the cathode rays. Now he changed the direction of the external magnetic field and found that the beam of electrons is deflected in the opposite direction. The magnetic field deflects the cathode ray. Now he applied a magnetic field across the cathode ray tube by using an external magnetic field. He had already deduced that the particles were negatively charged.įirstly, he applied an electric field in the path between anode and cathode and measured the deflections from the straight path. Then, he performed the third experiment to know the particles’ nature and reduce the particle’s mass as they had too small of a mass to be calculated directly.įor the experiment, he used the cathode ray tube with a high applied potential difference between the two electrodes, with the negatively charged cathode produced the cathode rays. This proved that the cathode rays were negatively charged. When the charged metal plates were introduced, he found that the cathode rays bent away from the negative plate and towards the positive plate. Instead of an electrometer at one end of the Cathode Ray Tube, he used a fluorescent-coated tube that would glow were the cathode ray hit it. He put a negatively charged metal plate on one side of the cathode rays to go past the anode and a positively charged metal plate on the other side. Then, he conducted a Second experiment to prove the charge carried by the cathode rays was negative or positive. He deduced that the electric charge and the cathode rays must be combined and are the same entity. The metal had two small diversions(slits), leading to an electrometer to measure small electric charges.įrom the first experiment, he discovered that the electrometers stopped measuring electric charge. He built his cathode ray tube with a metal cylinder on the other end. Thomson conducted his first cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. J.J.Thomson conducts three experiments with cathode ray experiment.
